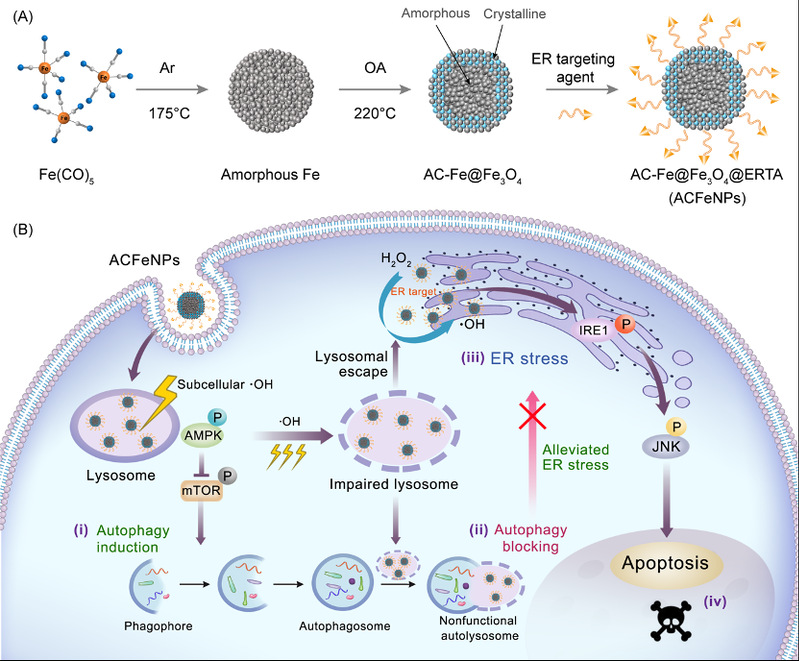
The spatiotemporal characterization of signaling crosstalk between subcellular organelles is crucial for the therapeutic effect of malignant tumors. Blocking interactive crosstalk in this fashion is significant but challenging. Herein, a communication interception strategy is reported, which blocks spatiotemporal crosstalk between subcellular organelles for cancer therapy with underlying molecular mechanisms. Briefly, amorphous-core@crystalline-shell Fe@Fe3O4 nanoparticles (ACFeNPs) are fabricated to specifically block the crosstalk between lysosomes and endoplasmic reticulum (ER) by hydroxyl radicals generated along with their trajectory through heterogeneous Fenton reaction. ACFeNPs initially enter lysosomes and trigger autophagy, then continuous lysosomal damage blocks the generation of functional autolysosomes, which mediates ER–lysosome crosstalk, thus the autophagy is paralyzed. Thereafter, released ACFeNPs from lysosomes induce ER stress. Without the alleviation by autophagy, the ER-stress-associated apoptotic pathway is fully activated, resulting in a remarkable therapeutic effect. This strategy provides a wide venue for nanomedicine to exert biological advantages and confers new perspective for the design of novel anticancer drugs.
Article link:https://onlinelibrary.wiley.com/doi/full/10.1002/adma.202211597





